RHINOMED TECHNOLOGY VS BASELINE
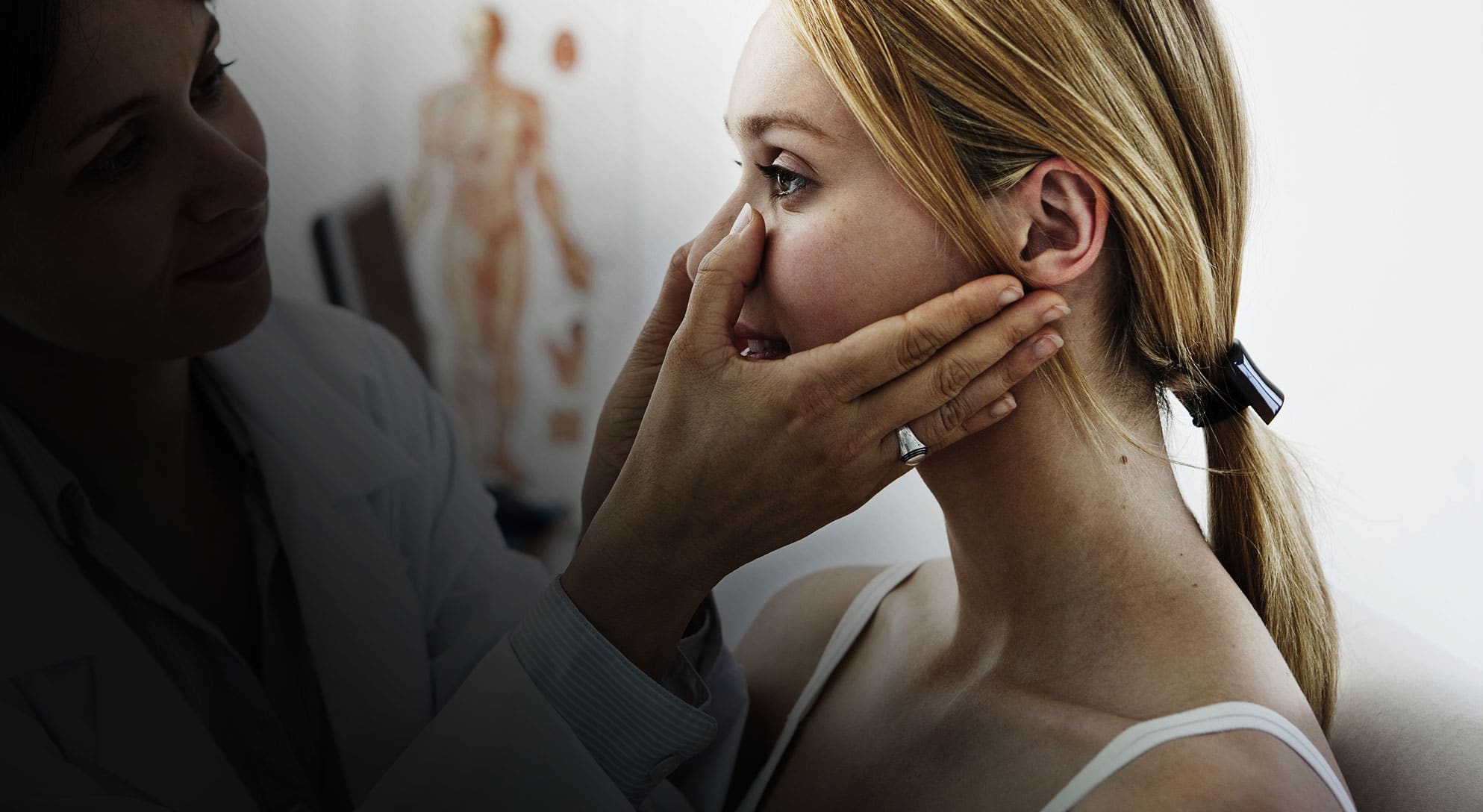
Clinical trials tested the effectiveness of the Turbine technology with impressive results.

Clinical trial
The Objective
Study the effect of nasal airflow with and without an intranasal stenting device*.
The Subjects
Group of twenty healthy individuals (male and female of varying ages).
The Test
Controlled Rhinometry test (participants mouths closed) where the amount of nasal airflow was measured using a Rhinomanometer.
The Findings
On average the intranasal stent provided 38% increase in airflow.
* Reference Braham, S Royal Melbourne Ear & Eye Hospital.
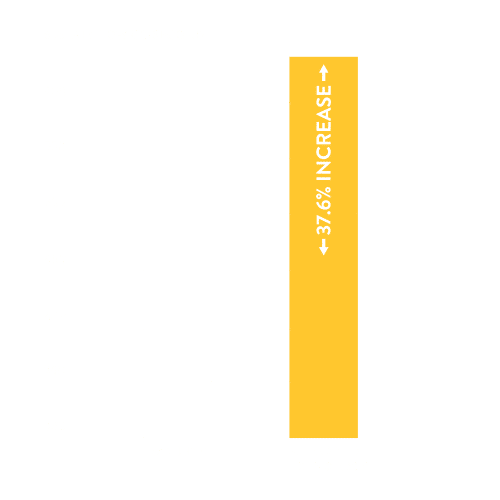

THE STUDY SHOWED
- Stenting the nasal valves via Rhinomed’s technology significantly improves airflow. The graph above shows a 37.6% increase in airflow over the baseline measure (p<0.05).
- Rhinomed’s technology is significantly better than baseline at improving airflow (p<0.05).
We knew from our earliest prototype that we were onto something great. So we put our earlier design head to head with baseline in a 2003 clinical trial to prove which would deliver the most airflow when tested under controlled, laboratory conditions.
Our experts know that fifty percent of the resistance in the nose is at the nasal valve, so all our devices target this area for stenting. The laws of physics support the hypothesis that even a small change to the opening of the valve, which at only 2.5mm wide is the narrowest part of the human airway, will have a dramatic improvement on airflow.
This theory of resistance is based on the formula that the airflow in a rigid tube (like the nasal cavity) is proportional to its radius to the power of four.

ABOUT THE PRODUCT
The early version of Rhinomed’s internal stenting technology, was made from an ultra soft polymer and was worn inside the nose to gently hold open each individual nostril, by an adjustable ratchet mechanism.
WHERE WE ARE TODAY?
Several years on from this study, our newest iteration of Rhinomed’s technology platform is Turbine™. The design has come along in leaps and bounds with features introduced specifically for sports use. Notably the shape now follows the contours of the internal nasal anatomy so it stays in place while you’re working out. The design team kept the independent dilation mechanism for a customised fit. Most importantly, as the nasal valve is key to increasing airflow as shown by our clinical trial, Turbine’s shape and position (when fitted correctly) precisely targets the valve making it a far superior product to other internal stents.
HOW WAS THE STUDY CONDUCTED?
The study was designed to validate and measure the increased nasal airflow that the Rhinomed’s technology could deliver, benchmarked against baseline.
In a randomised crossover study, twenty healthy volunteers underwent baseline assessment of their nasal flow rate and were then randomly assigned to test Rhinomed’s nasal stent or a competing device. Volunteers then crossed and tested the alternative device. Respondents were given at least ten minutes rest between testing each device and each of the three tests they undertook.
The principal investigator of the study, Dr Simon Braham, was a senior nasal surgeon at the Royal Victorian Eye and Ear Hospital, Melbourne Australia who has a special interest in treating the nasal valve. The study was performed at the Royal Victorian Eye and Ear hospital using a rhinomanometer, which is the gold standard tool to objectively measure pressure and flow through the nasal airway. Statistical analysis was performed by an independent statistician.
Buy turbine now
Buy Turbine from Rhinomed Shop. Choose your size now.



